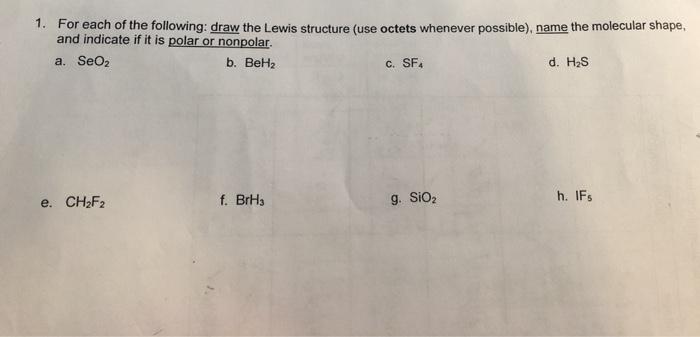
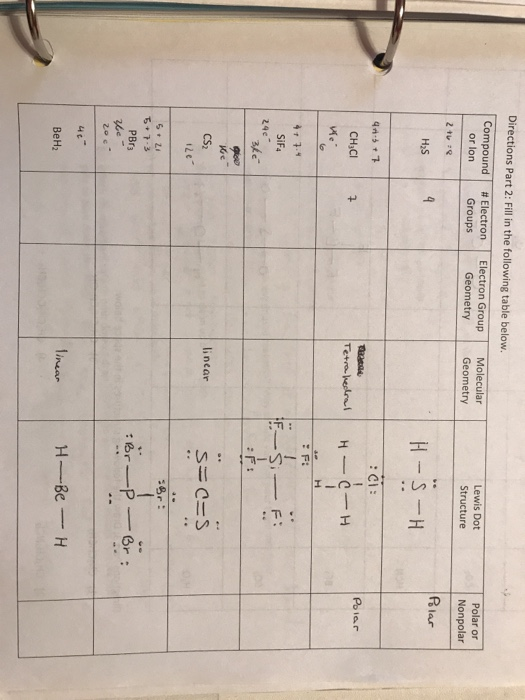
Write the electron configuration, Lewis structure, molecular geometry, and, electronic geometry for BeH_2. | Homework.Study.com
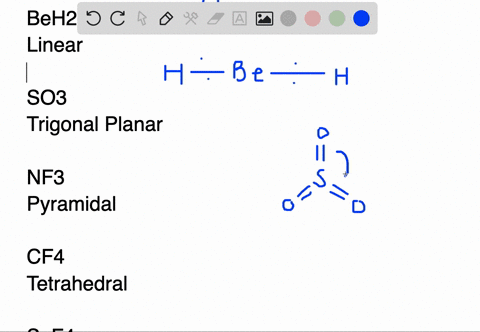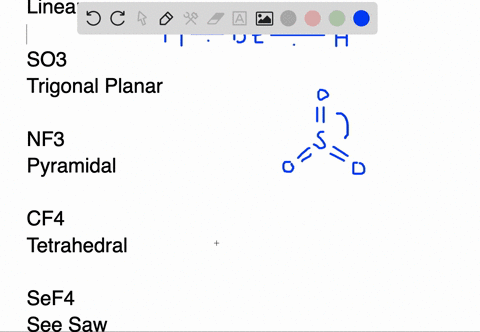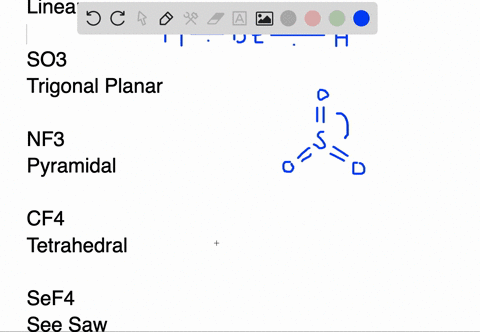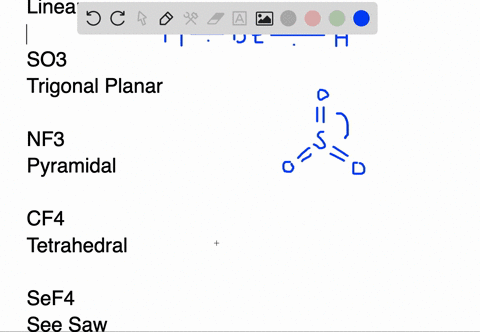
SOLVED:Draw Lewis structures and predict the molecular structures of the following. (See Exercises 89 and 90 . ) a. OCl2, KrF2, BeH2, SO2 c. CF4, SeF4, KrF4 b. SO3, NF3, IF3 d.

Which of the following molecules have the same geometries? a. CO2 and BeH2 b. SF4 and CH4 c. CO2 and NO2 d. N2O and NO2 | Homework.Study.com

BeH2 Lewis Structure (Beryllium Hydride) | BeH2 Lewis Structure (Beryllium Hydride) Beryllium Hydride or BeH2 has a simple arrangement of atoms. In this video, we help you find out the Lewis dot...

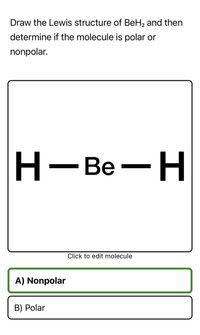
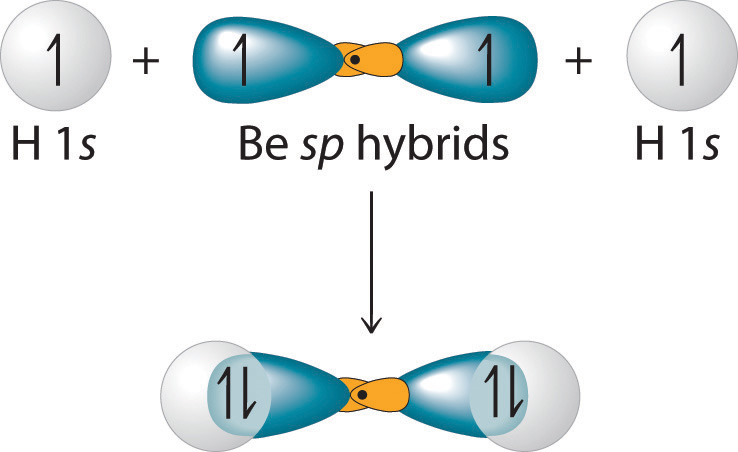
SOLVED:The bonds in beryllium hydride (BeH 2 ) molecules are polar, and yet the dipole moment of the molecule is zero. Explain.