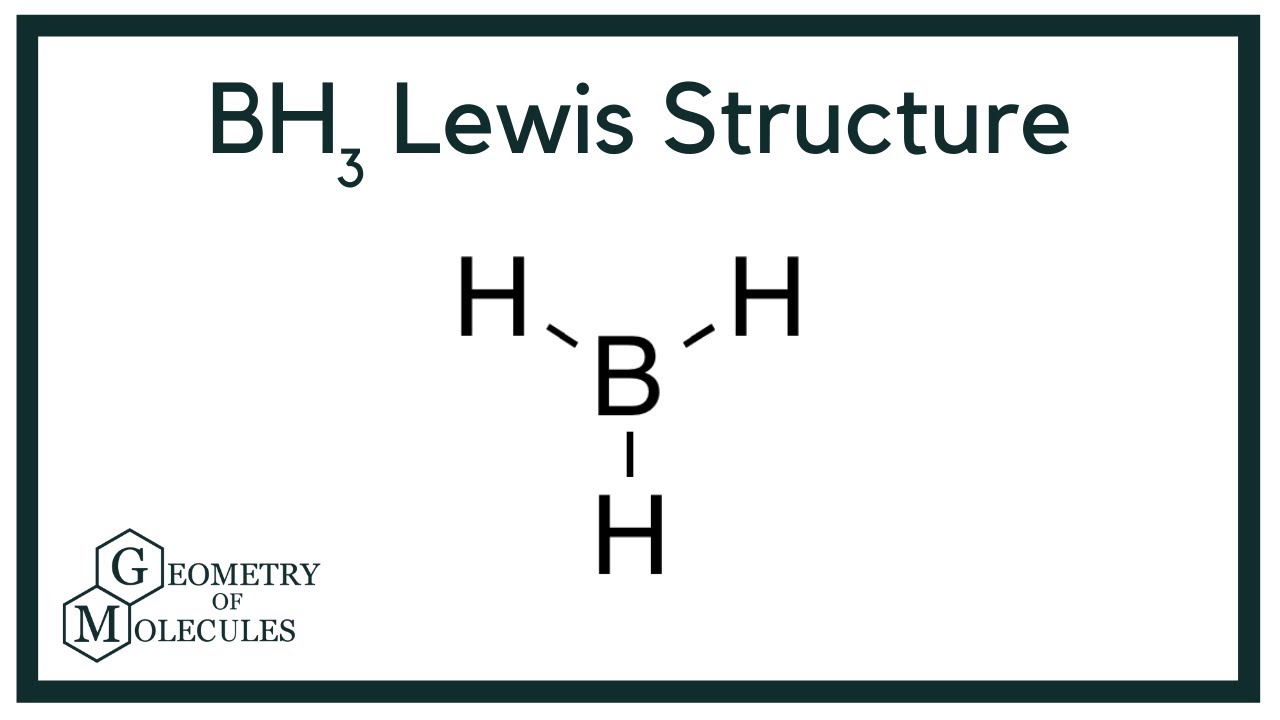
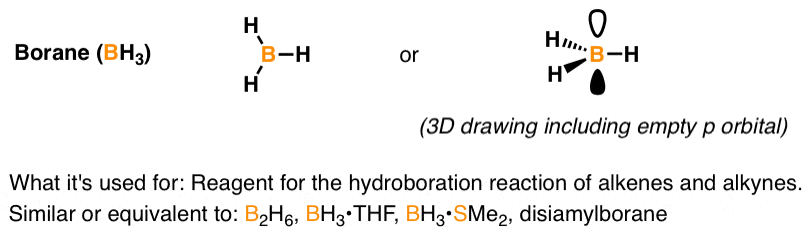
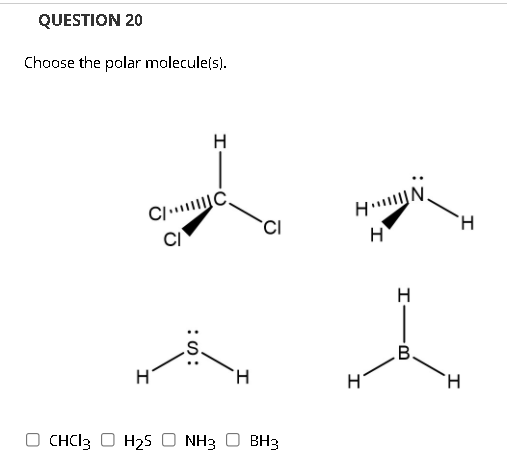
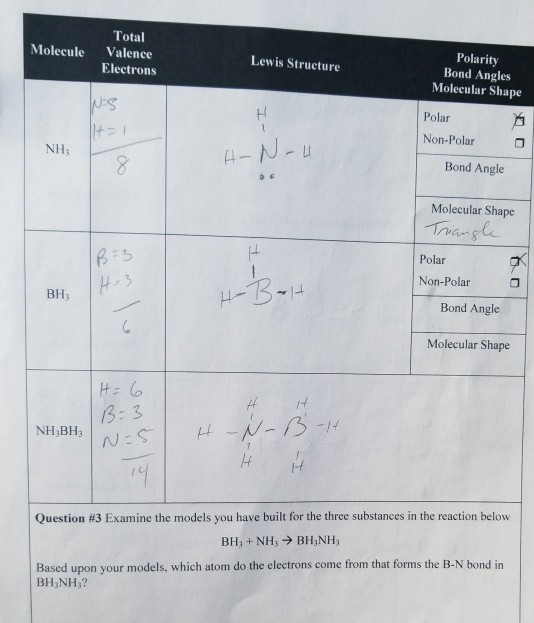
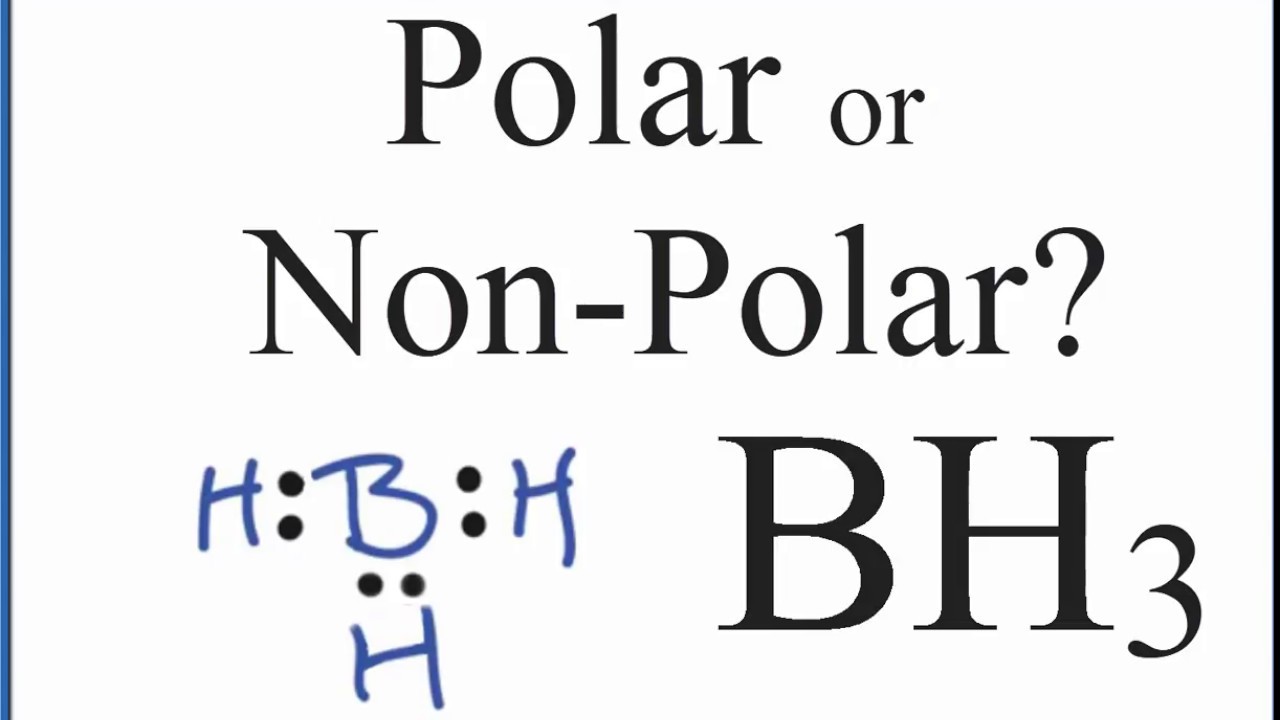SOLVED: uestion 8 Determine if this molecule (BH3) is polar or nonpolar H ot yet answered 'oints oul ol 1.00 Flag' question HB-H Select one: a, it cannot be determined b. nonpolar
1. What are the properties of IONIC substances? These substances: -Solid Hard and brittle (like salt) at room temp -Conduct electricity when dissolved. - ppt download