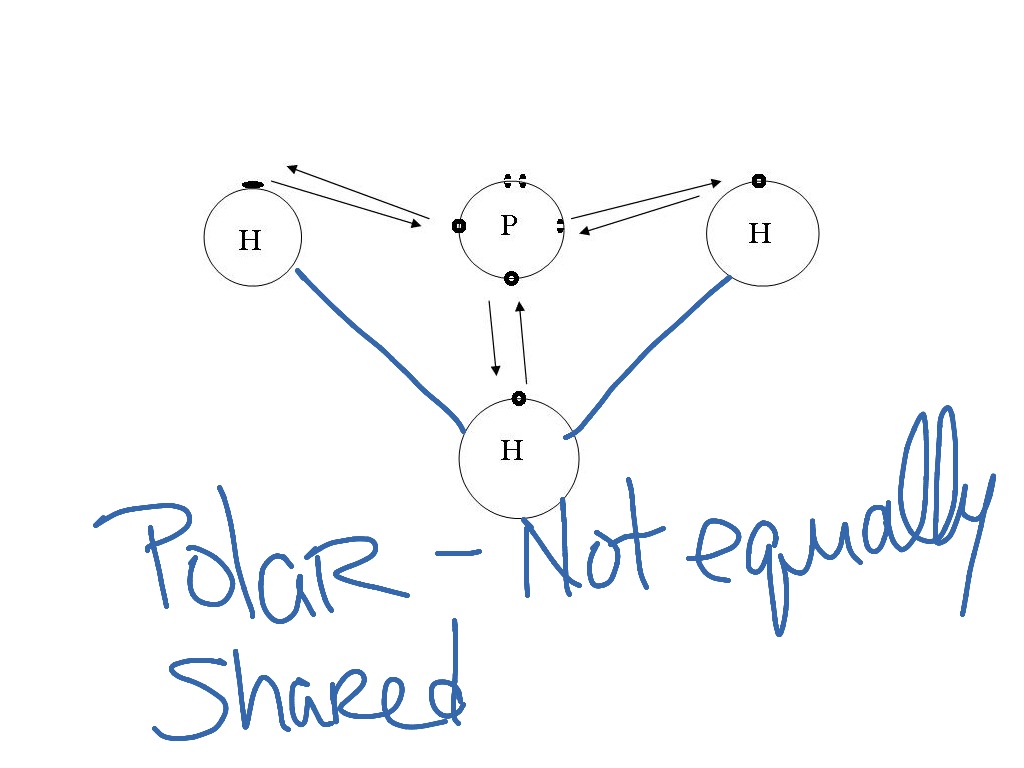
Circle the functional groups in the given compound and label these compounds as polar or nonpolar. Are they capable of Hydrogen bonding? | Homework.Study.com

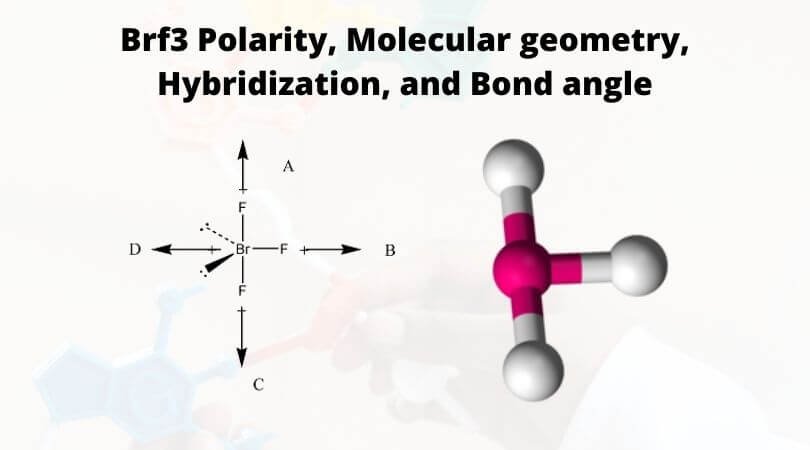
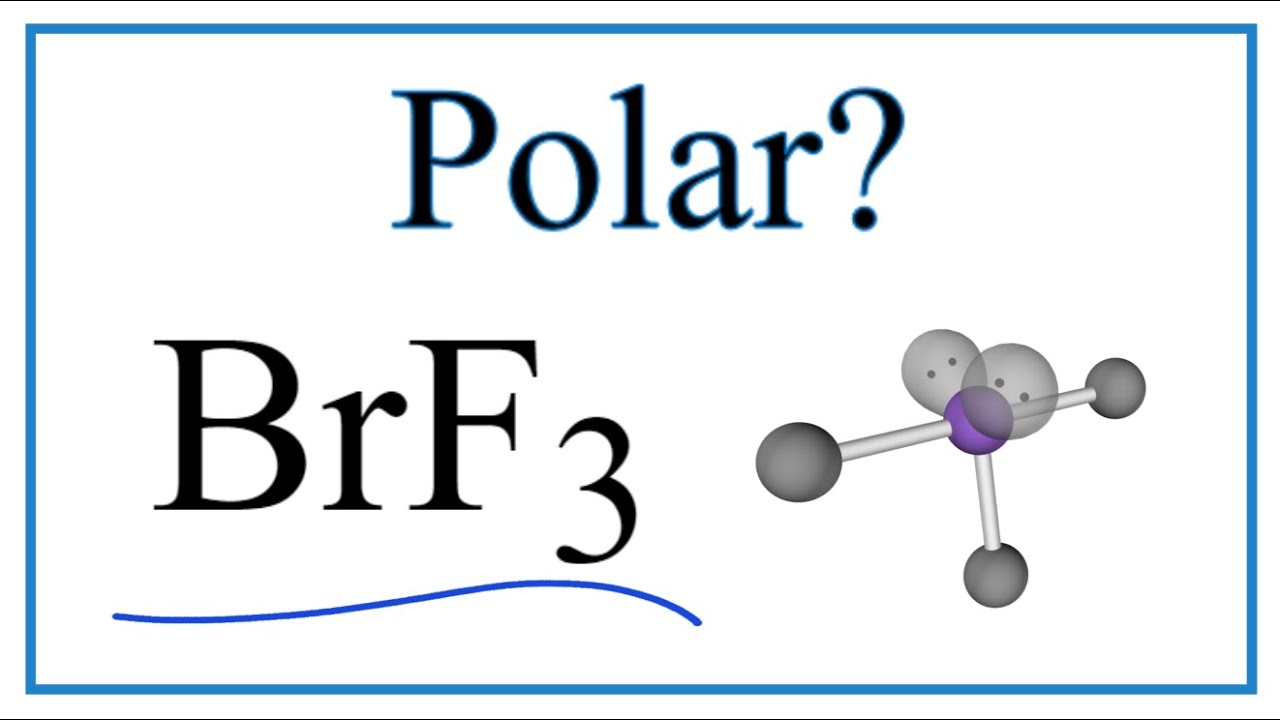
Which have a molecular dipole moment? (Select all that apply.) 1. BF3 2. SF4 3. BrF3 4. NF3 5. CF4 | Homework.Study.com