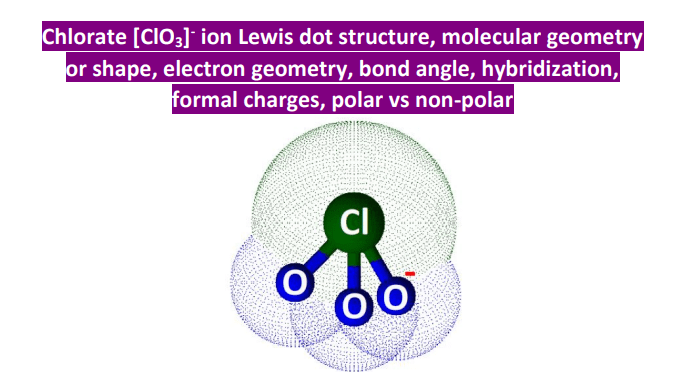
ClO3- Lewis Structure (Chlorate Ion) | ion, molecule | ClO3- Lewis Structure (Chlorate Ion) Here in this video, we will help you determine the ClO3- Lewis structure. Chlorate ion has a negative

ClO3- Molecular Geometry,Shape & Bond Angles (Chlorate Ion) Chlorate ion or ClO3- ion comprises one Chlorine atom that is in the center forming bonds... | By Geometry Of Molecules | Facebook

Draw the Lewis structure for ClO3- with all resonance forms. State its formal charge and polarity. What is its molecular geometry? | Homework.Study.com
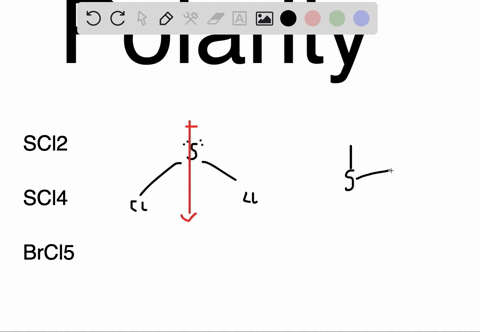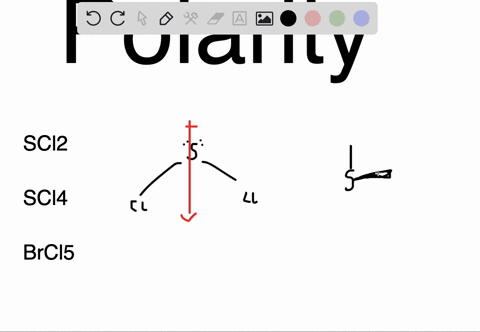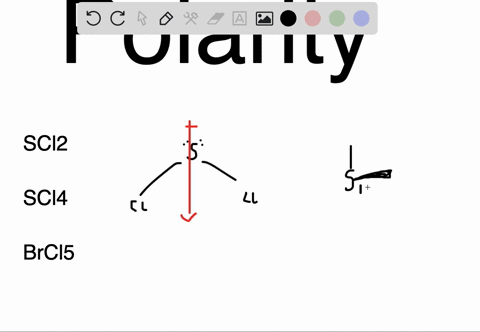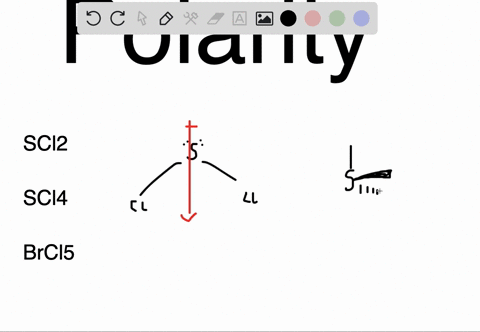
SOLVED: Determine whether each molecule or ion is polar or nonpolar. a. ClO3 - b. SCl2 c. SCl4 d. BrCl5

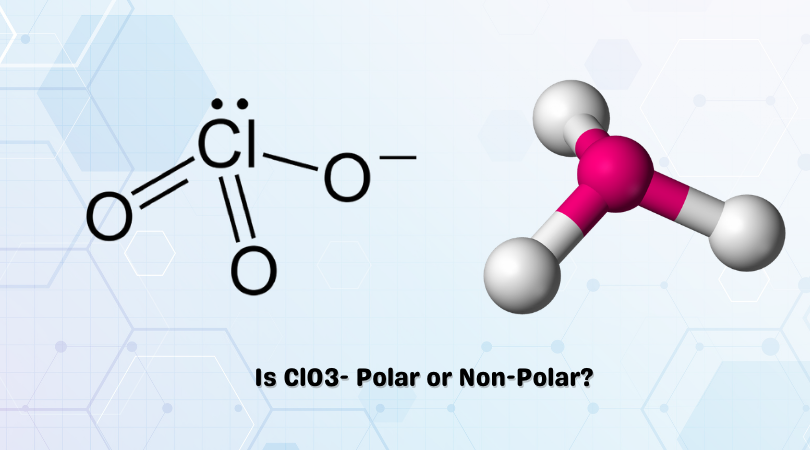
Science Coverage: Is ClO3- Polar or Nonpolar? | Molecular geometry, Covalent bonding, Oxidation state

SOLVED:Draw Lewis structures and give the other information requested for the following molecules: (a) BF3 Shape: planar or nonplanar? (b) ClO3^-. Shape: planar or nonplanar? (c) H2 O. Show the direction of

According to VSEPR theory, what values are expected for the O-Cl-O bond angles in the chlorate ion? | Homework.Study.com

SOLVED:Determine whether each molecule or ion is polar or nonpolar. a. ClO3 - b. SCl2 c. SCl4 d. BrCl5