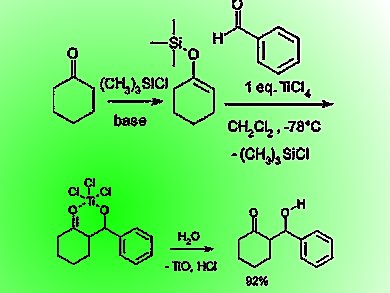
Diastereoselectivity in Lewis-Acid-Catalyzed Mukaiyama Aldol Reactions: A DFT Study | Journal of the American Chemical Society
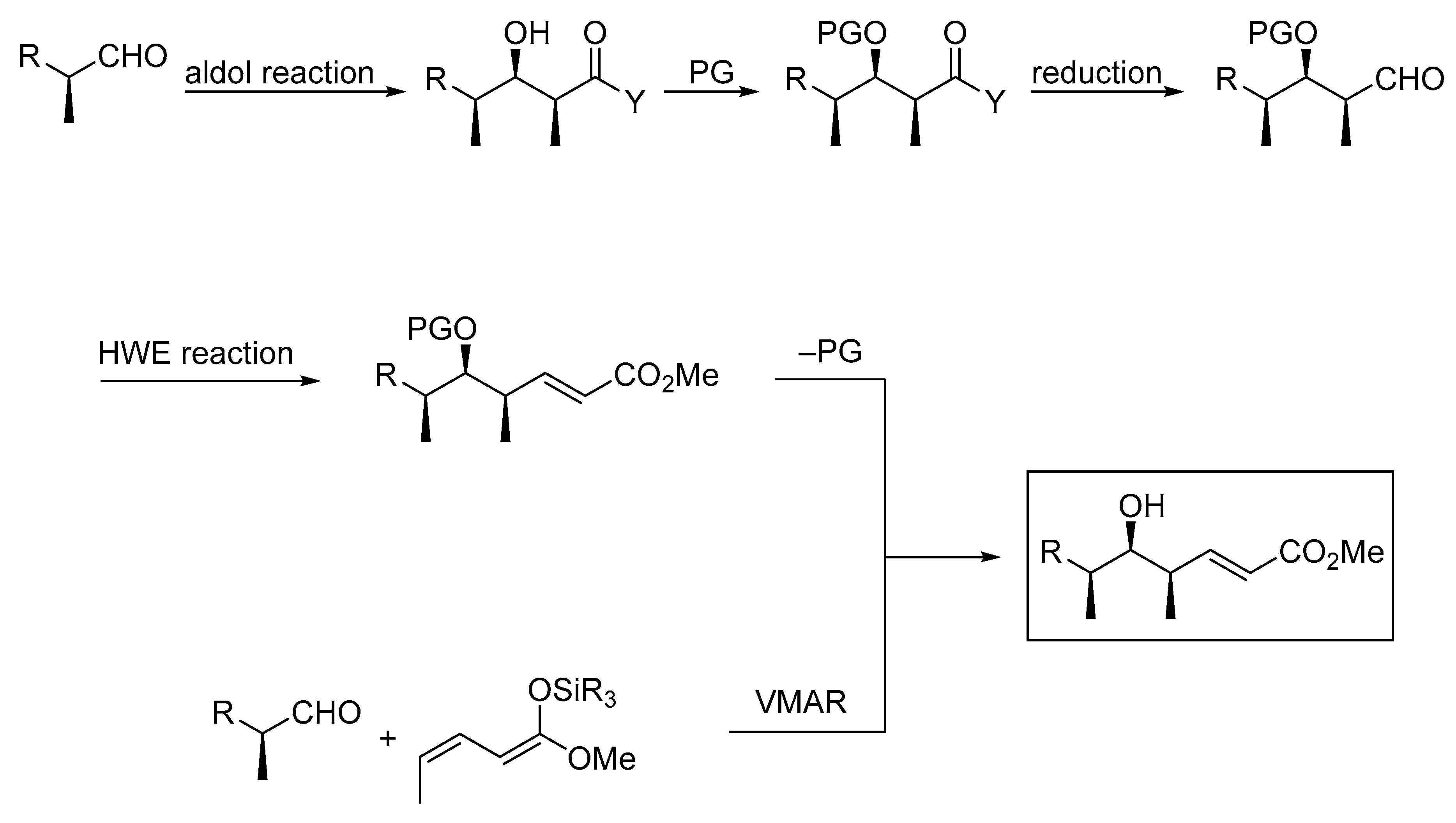
Molecules | Free Full-Text | Very Recent Advances in Vinylogous Mukaiyama Aldol Reactions and Their Applications to Synthesis

Mukaiyama Aldol Reactions in Aqueous Media - Kitanosono - 2013 - Advanced Synthesis & Catalysis - Wiley Online Library

Mukaiyama Aldol Reaction Catalyzed by (Benz)imidazolium-Based Halogen Bond Donors | Catalysis | ChemRxiv | Cambridge Open Engage

The Mukaiyama Aldol Reaction: 40 Years of Continuous Development - Matsuo - 2013 - Angewandte Chemie International Edition - Wiley Online Library

Diastereoselectivity in Lewis-acid-catalyzed Mukaiyama aldol reactions: a DFT study. | Semantic Scholar
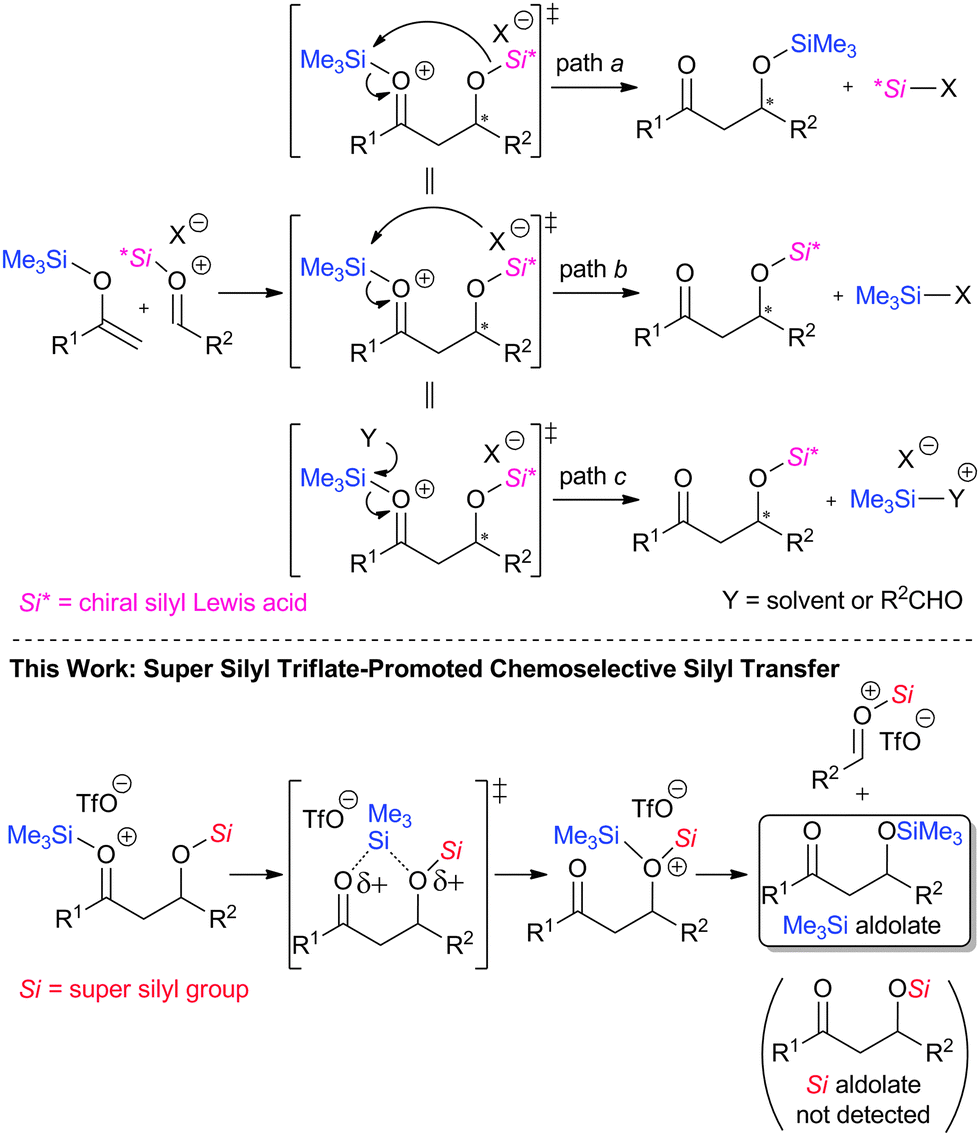
Chemoselective silyl transfer in the Mukaiyama aldol reaction promoted by super silyl Lewis acid - Chemical Communications (RSC Publishing) DOI:10.1039/C4CC05807K

The Mukaiyama Aldol Reaction: 40 Years of Continuous Development - Matsuo - 2013 - Angewandte Chemie International Edition - Wiley Online Library